Underlined are linked*
How Biotech Startup Funding Will Change in the Next 10 Years
If we start seeing decreased cost of R&D/commercialization, there will be an explosion of academics turning into founders and more startups in biotech. The example used is a company in the YC batch that runs all of its clinical trials in the cloud and is fully remote. Rather than company creation models dominating the space, we may start to see more founder led ventures.
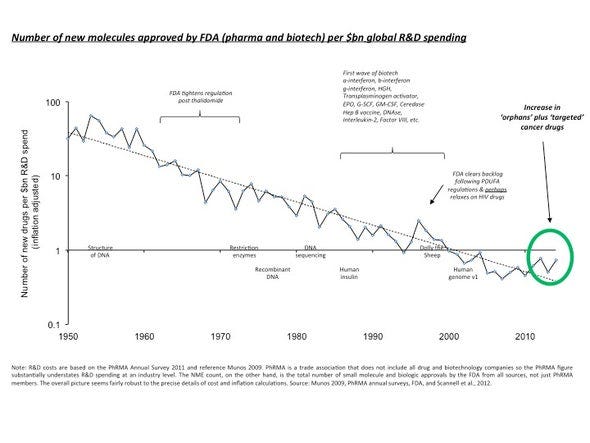
I have a general macro thesis that there will be a lot more startup founders in the years to come, and that biotech is one of the areas that will come to rise. We saw it in software, because anyone can learn to code in 6 months and build an app with operating leverage to scale to millions. That isn’t the case for biotech due to a variety of headwinds that I am hoping change in our lifetime: FDA and Eroom’s law (see image), the need for domain expertise and the difficulty of acquiring it, IP, university institutionalized research and the replicability crisis, etc. The FDA continues to increase the barrier of entry to company formation in biotech as innovation gets easier.
My hope is Moore’s law (semiconductors doubling every two years) overtakes Eroom’s law (cost doubling every 9 years) as … “biotech goes from analog to digital.” Robert Nelsen’s comparison to the rise of Moore’s law when computers started employing the current signal transmission mechanism. A part of a second macro thesis that AI and computation will bleed into every area of our world.
“Today, early stage biotech funding is dominated by the “venture creation model”. In the venture creation model, the VC firm creates the company. They have an initial idea and put together a team of favored executives, often from their pool of entrepreneurs-in-residence, to run it. The startup is typically incubated out of the VC’s offices. The VC invests a large amount of money upfront and takes a controlling ownership stake.
Just as VC-incubated tech companies made sense when tech companies were expensive to start, this model made sense when the cost to start a biotech company was high. Until recently, no one could get anything done before a VC wrote a $10M check, so this was the only way to get started.
But that’s no longer the case. Just like new infrastructure brought down the cost to start a tech company, new infrastructure has brought down the cost of doing biology dramatically. Today, founders can make real progress proving a concept for a biotech company for much less, often as little as $100K. There are low cost CROs that will do scientific work for a fee. Companies like Science Exchange make access to CROs and scientific supplies instantaneous and cost effective to small companies. It’s easy to rent fully equipped lab space by the bench, and there are companies to help you stock it. Affordable lab robots from companies like OpenTrons make it possible to automate batch experiments, and computational drug discovery from companies like Atomwise allows some experiments to be done completely in silico. Companies like Cognition IP are bringing down the cost of filing patents, and companies like Enzyme are streamlining FDA submission.”
Gene therapy has had its difficulties, and one of those well-known is the difficulty of delivery. LNPs, AAV, etc all have their challenges. Feng Zhang, Ph.D. who drive the application of CRISPR in mammalian systems out of MIT, tapped an Alnylam exec to launch Aera, with the hopes of developing a new protein-based delivery method. Backed by ARCH, Lux, and GV. If the $193 mm check size is any indication, the company may be onto something.
In connection with my first Y Combinator point, the venture launched not long after Dimension Capital launched with a similar purpose, both collectively raising $850 million. The funds both act as a microcosm to me that (1; Curie) there will be an increased focus on helping founders like YC and (2; Dimension) that we are entering the digital age of biotech looking to invest in computational startups.
I think probably first, Pharma became outdated and turned into a late-stage VC (money provider) essentially as they struggled to innovate, then the RA Capital model developed which took advantage of the massive opportunity in untapped university technology ready to be commercialized across the “valley of death” transition through clinical trials. Essentially offering company formation advisory, forming teams around the asset, and the infusion of cash to get them necessary runway. The final stage will probably look something like a Y Combinator, which seems to be the best seed venture model ever created, and is where Curie.bio is headed.
Robert Nelsen (conference talk with Josh Wolfe)
I don’t remember, just really like Nelsen as a guide point. He’s an outsider and thinks bigger than people who have been in the industry long enough to become numb to Medicine’s systemic stagnation.
Now researchers suggest it may be driving Alzheimer's disease. An ancient human foraging instinct, fueled by fructose production in the brain, may hold clues to the development and possible treatment of Alzheimer's disease (AD), according to researchers at the University of Colorado Anschutz Medical Campus. In this research, it seems scientists are thinking that there is the potential that prolonged exposure to Fructose basically eliminates the “fasted” state and the body can become adjusted to a state of abundance and the “survival state gets stuck in the ‘on’ position.” Doesn’t seem super fleshed out but I’ve heard the link between intermittent fasting and AD before.
“A study found that if you keep laboratory rats on fructose long enough, they get tau and amyloid beta proteins in the brain, the same proteins seen in Alzheimer’s disease,” Johnson said. “You can find high fructose levels in the brains of people with Alzheimer’s as well.”
Amyloid proteins are the same proteins that most of the companies that come across our desk and large anticipated drugs in AD are attempting to remove Amyloid like Aducanumab.
The fact that I don’t eat lunch is odd, most of my friends think so. But it is based in work out of David Sinclair’s lab on the Disposable Soma Theory of Aging, that essentially theorizes that the body has a tradeoff of repair and growth, and that the body wasn’t meant to have an abundance of food. In a fasted state, the body will engage in autophagy and clear up defective cells.
[NOTE]
When I was younger, I was “interested” in lots of types of innovation/tech. But I could never find a good source for anything and biotech specifically. Since coming into the job, I’ve been able to find a couple sources. Most of the information I gather in general comes from:
Hacker News – Y Combinator Forum for interesting things. Nerdy people tend to be interested in this website so cool things pop up, although it’s less often biotech and more software / startups oriented.
Fierce Biotech
WSJ
Equity research primers - Now that I’m starting to actually understand what I’m doing and the industry, these sources are interesting in going deeper into some companies’ technology
When you say send something that is the most interesting to me, that’s somewhat of a hard thing to unpack, and I think about why things interest me? Generally, the things that are interesting to me are those that teach me something, genuinely important news in an ongoing trend, or those that give some sort of signal into what the future holds--I don’t usually care about anything known. The problem with interesting things is they usually have a very dim signal. Lots of people talk about how they heard of bitcoin and the internet in their nascency and kick themselves for not investing, but it would have been difficult to determine how large those things would have been. You brought up Josh Wolfe yesterday, and I watched this video on him last night. He put it into words well: he reads broadly across news and journals like Science and Nature (may try to add these to my sources) and tries to pick up on dim signals. That’s what I’m going to start trying to do.